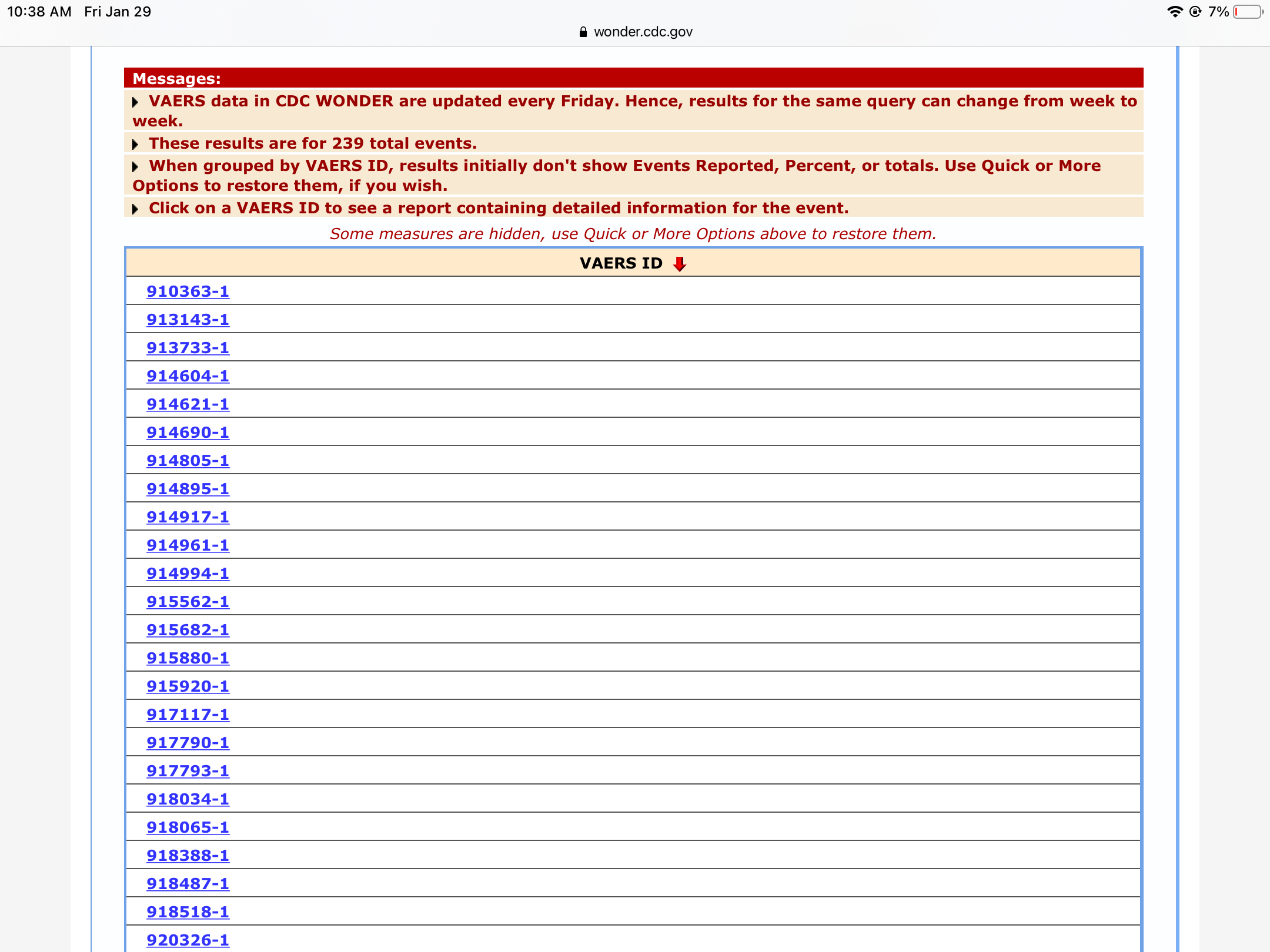

Only after additional research was evidence an association between the vaccine and intussusception found. After noticing the unusual number of reports, the RotaShield vaccine was removed from the market for further study. For example, in the 1999, VAERS detected more reports for intussusception after the RotaShield rotavirus vaccine that expected to occur by chance alone. What is the value of reports submitted to VAERS? Although reports submitted to VAERS are not evidence of vaccine side effects, the reports are used to identify potential adverse events. Reports submitted to VAERS cannot be used as evidence because the reports are unverified. The VAERS database contains information on unverified reports of adverse events such as illnesses, health problems, and symptoms following vaccination with US-licensed vaccines. The report of an adverse event to VAERS is not documentation that vaccine caused the event.” Therefore, VAERS collects data on any adverse event following vaccination, be it coincidental or truly caused by a vaccine. VAERS receives reports on all potential associations between vaccines and adverse events. “When evaluating data from VAERS, it is important to note that for any reported event, no cause-and-effect relationship has been established. In many cases, adverse events following vaccination are coincidental. In other words, just because an event occurred after the administration of a vaccine does not mean that the vaccine caused the event. Second, all adverse events submitted to VAERS are correlational. Knowingly filing a false VAERS report with the intent to mislead the Department of Health and Human Services is a violation of Federal law punishable by fine and imprisonment, but false reports are still likely submitted. For example, I can go to the VAERS website and submit my own report in a matter of minutes. Yes, anyone including health care providers, manufacturers, and vaccine recipients or their parents or guardians can submit a report. However, upon investigating the reporting system, some serious flaws emerge - flaws that mean that submitted reports are not evidence of vaccine side effects.įirst, anyone can submit a report to VAERS. Reports submitted to VAERS are monitored to detect new, unusual, or rare vaccine adverse events monitor increases in known adverse events identify potential patient risk factors for particular types of adverse events, identify vaccine lots with increased numbers or types of reported adverse events and assess the safety of newly licensed vaccines.Īt first glance, the VAERS database seems like a treasure trove of information about the side effects of vaccines. * Information listed above is at the time of submission.Approximately 30,000 VAERS reports are filed annually. This will benefit public health officials in that the availability of higher quality data and enhanced surveillance will allow for better informed public health decisions. The distribution and use of this application will increase the rate of reporting to the VAERS system and will also provide higher quality, and more complete data. Phase II will consist of preparing the application for broad distribution to healthcare providers. Phase I of this project entails the design and development of the application, and will serve as the proof of principal prototype. No other application currently exists to provide these needed capabilities. The application will also serve as a reference to healthcare providers for vaccine, vaccine safety, and other vaccination topics. The application will be capable of capturing all information required for submission to the Vaccine Adverse Event Reporting System (VAERS) and securely transmitting the information to the VAERS database.
This application will be targeted towards healthcare providers who make routine use of mobile devices in their daily work.
VAERS DATABASE ANDROID
A mobile computing application will be developed for Apple iOS and Google Android based mobile devices.
